Archive for the ‘Science News’ Category
Elon Musk Explains SpaceX’s Cutting-Edge Design Tech
Elon Musk, founder of SpaceX, explains some of the back-end technology his company is working on that will revolutionize the way engineering is done. First he shows the technology used to manipulate 3-D designs intuitively with a hand in 3-D space instead of in a clunky way with 2-D interfaces (like a mouse). Anyone who’s tried to do 3-D design in the usual software using a mouse and all sorts of buttons will appreciate the potential that this new interface represents.
Unfortunately, he only shows manipulation of the design – there’s no indication that they can actually design parts from scratch using this interface. As a consolation, though, we do get to see them printing rocket parts with a 3-D printer (you know, a replicator).
Even though it’s not the great leap I was hoping for when Musk tweeted about it as a teaser, it’s still a step in the right direction from a company that’s producing a startling number of those steps, so I couldn’t resist sharing the video:
Using 3-D Printers to Make Rocket Parts? Done.
Yes, according to this July 24th article on NASA’s website, not only can they manufacture injectors using 3-D printers at a substantially cheaper cost than by traditional methods, but the 3-D printed parts actually perform better. You can get all the juicy details from the NASA article – as usual, I’d like to talk about the broader implications. But first, how about a video of the part at work, enduring the 6,000 degree Fahrenheit temperatures it needs to:
Sometimes, NASA seems to be going at a snail’s pace that drives me nuts, but it looks like they’re on the cutting edge here, along with SpaceX, which is also using 3-D printers to make their parts.
Of course, every science fiction enthusiast has to be thinking replicators, right? And this is definitely going to be another case of science fiction becoming science fact, but let’s think about what this means for the next generation or so without getting all Star Trek about it. Think about Mars, or any future space base. Now, if something goes wrong, they won’t need the part manufactured on Earth and set on a potentially months-long delay. Instead, they’ll be taking a 3-D printer and some raw materials with them, and if they need to replace a part, they can manufacture it on-site without delay. This dramatically reduces the risks to any people we choose to send out there – especially for those sent to Mars.
Nanopollution in the Air
In an article a few weeks ago, I noted the way the synthetic particle cerium oxide had a tendency to stick around in incinerators, seeming indestructible. While worrying, this was not an immediate concern because cerium oxide is non-toxic, though it did suggest the need for more rigorous testing of all the chemicals we are putting into products – especially nano-scale synthetics that might pose a particular clean-up problem.
Well, research from Trinity College in Dublin might show that we’re already behind the curve on this. You can read the phys.org article on it here. Basically, the research showed that exposure carbon-based and silicon-based nanotechnology, including carbon nanotubes, can lead to rheumatoid arthritis and autoimmune diseases, as established by tests on human respiratory cells and on mice.
So, we now know that much of the new nanotechnology being developed may have serious health impacts. What we don’t know is where all the particles are going to end up or how easy or difficult it might be to clean it all up. Since many of the chemicals being developed are proprietary and treated as corporate secrets, there’s no guarantee that we’ll know what the new materials might be used in.
As the Guardian notes here, nanotechnology exists in a gaping hole in the regulatory framework, and companies are taking full advantage of the fact. Analysis of potential risks is virtually nonexistent. The question is, can university research money going to studies like that from Trinity College in Dublin keep up with the research and development money in the private sector. I think we know what the answer is.
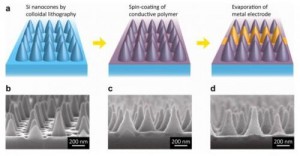
Nanocones for Cheap Solar Cells
This dovetails on the article about the solar-powered airplane. While the efficiency of solar panels is critical to making them worth deploying as an alternative to fossil fuels, that’s only half the issue. The other half, of course, is price. Right now, the cost is a dollar a watt, which is pretty hefty. Fortunately, increasing efficiency and decreasing price go hand-in-hand. In an earlier article, we introduced the idea that adding wrinkles might make the cells more efficient, and therefore cheaper. This time, it’s a different method of increasing surface area: cones.
Of course, because these are really small cones, they are dubbed “nanocones,” and the Stanford University study was published in “Nano Letters.” There will come a point where we will have to stop using nano- to prefix to everything that’s in the nanometer range, since most technology already incorporates components in that range, and in the near future, all of it will. We’ll need to be a bit more specific. After all, we already have carbon nanocones, which are altogether different in application from these silicon cones. But that’s just a pet-peeve of mine – for now, nano’s captured everyone’s imagination, so I guess we’ll go with it everywhere we can.
The key to the cost-savings of this design is the space between the cones. In a normal polymer cell, you need a full second layer for the polymer, but with this design, it can simply fill the gaps. Having a second layer involves using other materials, so this method renders those unnecessary.
Solar Airplane Upgrading For World Tour
Yesterday, I wrote an article about the energy we could get out of a fusion reactor in development. Today, it’s about the energy we could get from a fusion reactor already in service: the Sun.
Solar power lacks the flair of fusion, and I’m afraid the story of a plane that cruises at 43 mph probably won’t do much to help that along, but there’s a lot to the Solar Impulse that captures the imagination. There’s its magnificent wingspan, for instance:
That’s a 208 feet wingspan folks, dwarfing many airliners and bombers. The kicker is that the whole thing weighs in at a max takeoff weight of two tons – 4,400 pounds. It’s empty weight is about the same as your car’s. Think for a moment about the brilliant design and fine construction keeping the weight down must have required. There’s no two ways about it – just the size-to-weight issue makes this a marvel of engineering.
A Step Forward for Fusion Power
Could it be that, in my lifetime, we will at least develop viable fusion power? I mean, the various space agencies are killing my hope that we’ll send someone to Mars in the next fifty years, but maybe we’re all desperate enough for energy that we can pull this colossal trick off, and catapult civilization to the next stage?
University of Tennessee researchers have successfully tested the technology that keeps the central solenoid of a tokamak fusion reactor stable. The actual solenoid for the ITER (International Thermonuclear Experimental Reactor) in France can now be built by General Atomics in San Diego. If successful, ITER will start running deuterium-tritium reactions in 2026. The expectation is that it will be able to output ten times more power than it inputs. Considering fusion reactors up till now have consumed more than they produce, this is a big leap.
Just to orient ourselves, this is a tokamak-type reactor from the outside:
And the internal torus surrounding the solenoid:
On the Persistence of Nano-waste
First of all, a clarification. The prefix nano- seems to get attached to all sorts of stuff these days, especially in the science press, and it’s just begging for confusion. Nano-waste simply refers to synthetic particles on the nanometer scale that are engineered for some purpose, and are discarded along with the product when it has outlived its usefulness. It is nanotechnology in the broadest sense of the term – and we really need to be clearer about what we mean by nanotechnology going forward – though the conclusions reached by Walser, T. et. al. in the journal Nature Nanotechnology certainly has relevance to applications of small-scale computing (what we normally think about when we discuss nanotech) as well.
The particular problem raised by Walser, et al is that cerium oxide, a non-toxic synthetic used in catalytic converters, neither burns up nor biodegrades, so that it just sticks around and may eventually end up in our water supply and food cycle. Try to send it through an incinerator, and it will end up with the residue and tossed into a landfill, where it will persist indefinitely.
The good news is that cerium oxide is non-toxic and doesn’t escape into the atmosphere through the incineration process. The bad news is that it is not the only synthetic we have or are developing, and in many cases the environmental impact of the new compounds is not adequately tested. Once researchers find a practical use for them, they’re put to work without regard for whether we can get rid of them, or what they might do to us once they end their useful life and wind up in a pile of waste.